The POWER Procedure
- Overview
-
Getting Started

-
Syntax

-
Details

-
Examples
 One-Way ANOVAThe Sawtooth Power Function in Proportion AnalysesSimple AB/BA Crossover DesignsNoninferiority Test with Lognormal DataMultiple Regression and CorrelationComparing Two Survival CurvesConfidence Interval PrecisionCustomizing PlotsBinary Logistic Regression with Independent PredictorsWilcoxon-Mann-Whitney Test
One-Way ANOVAThe Sawtooth Power Function in Proportion AnalysesSimple AB/BA Crossover DesignsNoninferiority Test with Lognormal DataMultiple Regression and CorrelationComparing Two Survival CurvesConfidence Interval PrecisionCustomizing PlotsBinary Logistic Regression with Independent PredictorsWilcoxon-Mann-Whitney Test - References
Example 71.10 Wilcoxon-Mann-Whitney Test
Consider a hypothetical clinical trial to treat interstitial cystitis (IC), a painful, chronic inflammatory condition of the bladder with no known cause that most commonly affects women. Two treatments will be compared: lidocaine alone (“lidocaine”) versus lidocaine plus a fictitious experimental drug called Mironel (“Mir+lido”). The design is balanced, randomized, double-blind, and female-only. The primary outcome is a measure of overall improvement at week 4 of the study, measured on a seven-point Likert scale as shown in Table 71.34.
Table 71.34: Self-Report Improvement Scale
|
Compared to when I started |
|
|
this study, my condition is: |
|
|
Much worse |
–3 |
|
Worse |
–2 |
|
Slightly worse |
–1 |
|
The same |
0 |
|
Slightly better |
+1 |
|
Better |
+2 |
|
Much better |
+3 |
The planned data analysis is a one-sided Wilcoxon-Mann-Whitney test with ![]() where the alternative hypothesis represents greater improvement for “Mir+lido.”
where the alternative hypothesis represents greater improvement for “Mir+lido.”
You are asked to graphically assess the power of the planned trial for sample sizes between 100 and 250, assuming that the conditional outcome probabilities given treatment are equal to the values in Table 71.35.
Table 71.35: Conjectured Conditional Probabilities
|
Response |
|||||||
|---|---|---|---|---|---|---|---|
|
Treatment |
–3 |
–2 |
–1 |
0 |
+1 |
+2 |
+3 |
|
Lidocaine |
0.01 |
0.04 |
0.20 |
0.50 |
0.20 |
0.04 |
0.01 |
|
Mir+lido |
0.01 |
0.03 |
0.15 |
0.35 |
0.30 |
0.10 |
0.06 |
Use the following statements to compute the power at sample sizes of 100 and 250 and generate a power curve:
ods listing style=htmlbluecml;
ods graphics on;
proc power;
twosamplewilcoxon
vardist("lidocaine") = ordinal ((-3 -2 -1 0 1 2 3) :
(.01 .04 .20 .50 .20 .04 .01))
vardist("Mir+lido") = ordinal ((-3 -2 -1 0 1 2 3) :
(.01 .03 .15 .35 .30 .10 .06))
variables = "lidocaine" | "Mir+lido"
sides = u
ntotal = 100 250
power = .;
plot step=10;
run;
ods graphics off;
The ODS LISTING STYLE=HTMLBLUECML statement specifies the HTMLBLUECML style, which is suitable for use with PROC POWER because it allows both marker symbols and line styles to vary. See the section ODS Styles Suitable for Use with PROC POWER for more information.
The VARDIST= option is used to define the distribution for each treatment, and the VARIABLES= option specifies the distributions to compare. The SIDES=U option corresponds to the alternative hypothesis that the second distribution ("Mir+lido") is more favorable. The NTOTAL= option specifies the total sample sizes of interest, and the POWER= option with a missing value (.) identifies the parameter to solve for. The default GROUPWEIGHTS= and ALPHA= options specify a balanced design and significance level ![]() .
.
The STEP=10 option in the PLOT statement requests a point for each sample size increment of 10. The default values for the X=, MIN=, and MAX= plot options specify a sample size range of 100 to 250 (the same as in the analysis) for the X axis.
The tabular and graphical results are shown in Output 71.10.1 and Output 71.10.2, respectively.
Output 71.10.1: Power Values for Wilcoxon-Mann-Whitney Test
| Fixed Scenario Elements | |
|---|---|
| Method | O'Brien-Castelloe approximation |
| Number of Sides | U |
| Group 1 Variable | lidocaine |
| Group 2 Variable | Mir+lido |
| Pooled Number of Bins | 7 |
| Alpha | 0.05 |
| Group 1 Weight | 1 |
| Group 2 Weight | 1 |
| NBins Per Group | 1000 |
| Computed Power | ||
|---|---|---|
| Index | N Total | Power |
| 1 | 100 | 0.651 |
| 2 | 250 | 0.939 |
Output 71.10.2: Plot of Power versus Sample Size for Wilcoxon Power Analysis
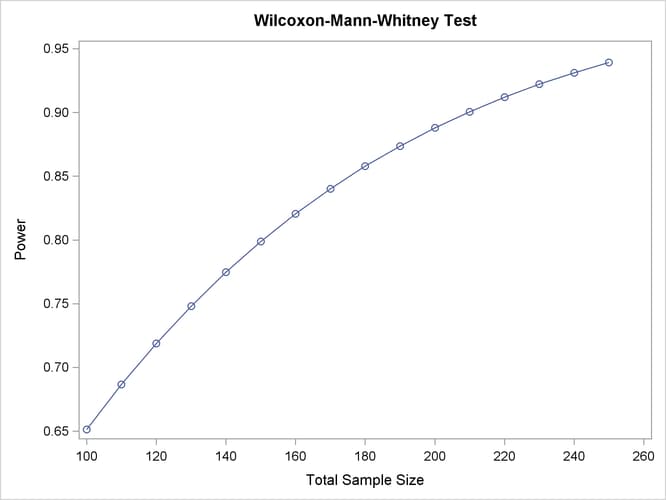
The achieved power ranges from 0.651 to 0.939, increasing with sample size.