| The SEQDESIGN Procedure |
| Applicable Two-Sample Tests and Sample Size Computation |
The SEQDESIGN procedure provides sample size computation for two-sample tests: the test for the difference between two normal means, tests for binomial proportions, and the log-rank test for two survival distributions. These tests for binomial proportions include the test for the difference between two binomial proportions, the log odds ratio test for binomial proportions, and the log relative risk test for binomial proportions,
For a test of difference between two sample means, the required sample size depends on the assumed sample variances. Similarly, for a test of two-sample proportions, the required sample size depends on the assumed sample proportions. For a log-rank test of two survival distributions, the required sample size depends on the assumed sample hazard rates, accrual rate, and accrual time.
If the REF=NULLPROP or REF=NULLHAZARD option is specified, the proportions or hazard rates under the null hypothesis are used to derive the required sample size or number of events. Otherwise, the REF=PROP option (which is the default in the MODEL=TWOSAMPLEFREQ option) or the REF=HAZARD option (which is the default in the MODEL=TWOSAMPLESURVIVAL option) uses proportions or hazard rates under the alternative hypothesis to derive the required sample size or number of events.
Test for the Difference between Two Normal Means
The MODEL=TWOSAMPLEMEAN option in the SAMPLESIZE statement derives the sample size required to test the difference between the means of two normal populations 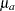 and
and 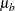 by using the null hypothesis
by using the null hypothesis 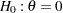 , where
, where 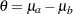 .
.
At stage 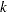 , the MLE for
, the MLE for 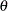 is computed as
is computed as
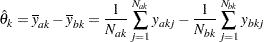 |
where 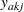 and
and 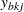 are the values of the
are the values of the  th observation available in the
th observation available in the 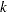 th stage groups A and B, respectively, and
th stage groups A and B, respectively, and 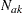 and
and 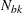 are the cumulative sample sizes at stage
are the cumulative sample sizes at stage 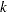 for these two groups.
for these two groups.
The statistic 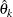 has a normal distribution
has a normal distribution
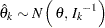 |
where the information 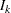 is the inverse of the variance
is the inverse of the variance 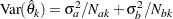 .
.
Then the standardized statistic
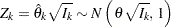 |
Thus, to test the hypothesis 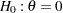 against an upper alternative
against an upper alternative 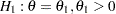 ,
, 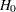 is rejected at stage
is rejected at stage 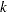 if the statistic
if the statistic 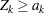 , the upper
, the upper  boundary for the standardized
boundary for the standardized 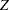 statistic at stage
statistic at stage 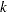 .
.
If the variances  and
and 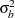 are unknown, the sample variances can be used to derive the information
are unknown, the sample variances can be used to derive the information 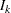 if it is assumed that each sample variance is computed from a large sample such that the test statistic has an approximately normal distribution.
if it is assumed that each sample variance is computed from a large sample such that the test statistic has an approximately normal distribution.
The maximum information is needed to derive the required sample size. If the maximum information is not specified or derived in the procedure, the alternative reference 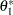 specified in the MEANDIFF option is used to derive the maximum information.
specified in the MEANDIFF option is used to derive the maximum information.
Note that in order to derive the sample sizes 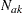 and
and 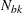 uniquely from the information,
uniquely from the information, 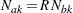 is assumed for
is assumed for 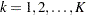 , where
, where  is the constant allocation ratio computed from the WEIGHT=
is the constant allocation ratio computed from the WEIGHT=
 option in the SAMPLESIZE statement.
option in the SAMPLESIZE statement.
In PROC SEQDESIGN, the computed total sample sizes for the two groups are
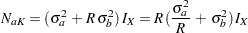 |
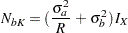 |
where 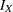 is the maximum information derived in the SEQDESIGN procedure,
is the maximum information derived in the SEQDESIGN procedure, 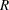 is the constant allocation ratio, and
is the constant allocation ratio, and 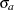 and
and 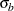 are the specified standard deviations.
are the specified standard deviations.
For 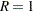 , the two sample sizes are equal, then
, the two sample sizes are equal, then
 |
If the variances from the two groups are equal, 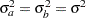 , then the total sample sizes for the two groups are
, then the total sample sizes for the two groups are
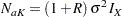 |
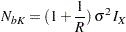 |
and the total sample size is
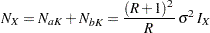 |
Furthermore, for 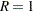 , the two sample sizes are equal, then
, the two sample sizes are equal, then
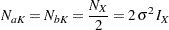 |
With an available maximum information, you can specify the MODEL=TWOSAMPLEMEAN( WEIGHT= 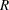 STDDEV=
STDDEV= 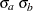 ) option in the SAMPLESIZE statement to compute the required total sample size and individual sample size at each stage. A procedure such as PROC GLM can be used to derive the two-sample
) option in the SAMPLESIZE statement to compute the required total sample size and individual sample size at each stage. A procedure such as PROC GLM can be used to derive the two-sample  test for the mean difference.
test for the mean difference.
Test for the Difference between Two Binomial Proportions
The MODEL=TWOSAMPLEFREQ(TEST=PROP) option in the SAMPLESIZE statement derives the sample size required to test the difference between two binomial populations with 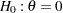 , where
, where 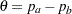 . At stage
. At stage 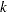 , the MLE for
, the MLE for 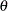 is
is
 |
where 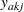 and
and 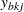 are the values of the
are the values of the  th observation available in the
th observation available in the 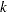 th stage for groups A and B, respectively, and
th stage for groups A and B, respectively, and 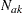 and
and 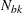 are the cumulative sample sizes at stage
are the cumulative sample sizes at stage 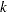 for these two groups.
for these two groups.
For sufficiently large sample sizes 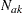 and
and 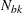 , the statistic
, the statistic 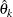 has an approximate normal distribution
has an approximate normal distribution
 |
where the information is the inverse of the variance
 |
Thus, the standardized statistic
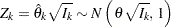 |
In practice, 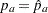 and
and 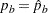 , the estimated sample proportions for groups A and B, respectively, at stage
, the estimated sample proportions for groups A and B, respectively, at stage 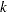 , can be used to derive the information
, can be used to derive the information 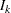 and the test statistic
and the test statistic 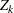 . Thus, to test the hypothesis
. Thus, to test the hypothesis 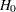 against an upper alternative
against an upper alternative 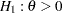 ,
, 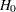 is rejected at stage
is rejected at stage 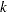 if the statistic
if the statistic 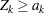 , the upper
, the upper  boundary for the standardized
boundary for the standardized 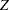 statistic at stage
statistic at stage 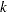 .
.
The maximum information 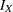 is needed to derive the required sample size. If the maximum information is not specified or derived with the ALTREF= option in the procedure, the PROP= option in the SAMPLESIZE statement is used to provide proportions under the alternative hypothesis for the alternative reference and then to derive the maximum information.
is needed to derive the required sample size. If the maximum information is not specified or derived with the ALTREF= option in the procedure, the PROP= option in the SAMPLESIZE statement is used to provide proportions under the alternative hypothesis for the alternative reference and then to derive the maximum information.
The proportions in the two groups are needed to derive the sample size. Also, in order to derive the sample sizes 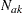 and
and 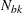 uniquely from the information,
uniquely from the information, 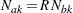 is assumed for
is assumed for 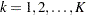 , where
, where  is the constant allocation ratio computed from the WEIGHT=
is the constant allocation ratio computed from the WEIGHT=
 option in the SAMPLESIZE statement. Then
option in the SAMPLESIZE statement. Then
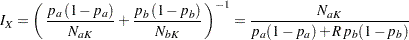 |
In PROC SEQDESIGN, the total sample sizes in the two groups are computed as
 |
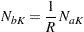 |
where  is the constant allocation ratio, and
is the constant allocation ratio, and 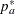 and
and 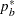 are proportions specified with the REF= option:
are proportions specified with the REF= option:
REF=NULLPROP uses proportions under
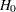 :
:  ,
, 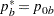
REF=AVGNULLPROP uses the average proportion under
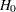 :
: 
REF=PROP uses proportions under
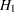 :
: 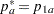 ,
, 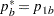
REF=AVGPROP uses the average proportion under
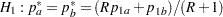
The total sample size is given by
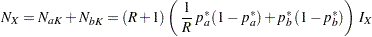 |
For 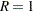 , the two sample sizes are equal,
, the two sample sizes are equal,
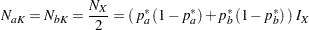 |
You can specify the MODEL=TWOSAMPLEFREQ( TEST=PROP WEIGHT= ) option in the SAMPLESIZE statement to compute the required total sample size and individual sample size at each stage. A procedure such as PROC GENMOD with the default DIST=NORMAL option in the MODEL statement can be used to derive the two-sample
) option in the SAMPLESIZE statement to compute the required total sample size and individual sample size at each stage. A procedure such as PROC GENMOD with the default DIST=NORMAL option in the MODEL statement can be used to derive the two-sample  test for proportion difference.
test for proportion difference.
Test for Two Binomial Proportions with a Log Odds Ratio Statistic
The MODEL=TWOSAMPLEFREQ(TEST=LOGOR) option in the SAMPLESIZE statement derives the sample size required to test two binomial proportions by using a log odds ratio statistic. The odds ratio is the ratio of the odds in one group to the odds in the other group, and the log odds ratio is the logarithm of the odds ratio
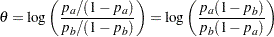 |
The hypothesis of no difference between two proportions, 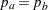 , can be tested through the null hypothesis
, can be tested through the null hypothesis 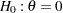 , where
, where 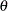 is the log odds ratio. For example, with
is the log odds ratio. For example, with 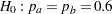 and
and  , it corresponds to the equivalent hypothesis
, it corresponds to the equivalent hypothesis 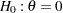 and
and 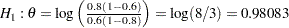 .
.
The maximum likelihood estimate of 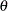 is given by
is given by
 |
with an asymptotic variance
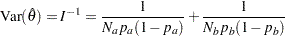 |
where 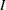 is the information (Diggle et al. 2002, pp. 341–342). That is, the standardized statistic
is the information (Diggle et al. 2002, pp. 341–342). That is, the standardized statistic
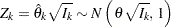 |
In practice, 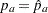 and
and 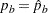 , the estimated sample proportions for groups A and B, respectively, at stage
, the estimated sample proportions for groups A and B, respectively, at stage 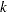 , can be used to derive the information
, can be used to derive the information 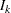 and the test statistic
and the test statistic 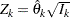 if the two sample sizes
if the two sample sizes 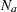 and
and 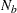 are sufficiently large such that the test statistic has an approximately normal distribution.
are sufficiently large such that the test statistic has an approximately normal distribution.
The maximum information 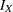 is needed to derive the required sample size. If the maximum information is not specified or derived with the ALTREF= option in the procedure, the PROP= option in the SAMPLESIZE statement is used to provide proportions under the alternative hypothesis for the alternative reference and then to derive the maximum information.
is needed to derive the required sample size. If the maximum information is not specified or derived with the ALTREF= option in the procedure, the PROP= option in the SAMPLESIZE statement is used to provide proportions under the alternative hypothesis for the alternative reference and then to derive the maximum information.
In order to derive the sample sizes 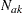 and
and 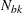 uniquely from the information,
uniquely from the information, 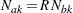 is assumed for
is assumed for 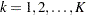 , where
, where  is the constant allocation ratio computed from the WEIGHT=
is the constant allocation ratio computed from the WEIGHT=
 option in the SAMPLESIZE statement. Then with
option in the SAMPLESIZE statement. Then with
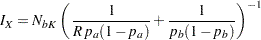 |
the sample size can be computed.
In PROC SEQDESIGN, the total sample sizes in the two groups are computed as
 |
 |
where  is the constant allocation ratio, and
is the constant allocation ratio, and 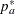 and
and 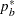 are proportions specified with the REF= option:
are proportions specified with the REF= option:
REF=NULLPROP uses proportions under
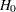 :
:  ,
, 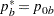
REF=AVGNULLPROP uses the average proportion under
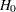 :
: 
REF=PROP uses proportions under
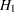 :
: 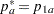 ,
, 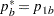
REF=AVGPROP uses the average proportion under
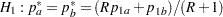
You can specify the MODEL=TWOSAMPLEFREQ( TEST=LOGOR WEIGHT=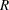 ) option in the SAMPLESIZE statement to compute the required total sample size and individual sample size at each stage. A procedure such as PROC LOGISTIC can be used to derive the log odds ratio statistic.
) option in the SAMPLESIZE statement to compute the required total sample size and individual sample size at each stage. A procedure such as PROC LOGISTIC can be used to derive the log odds ratio statistic.
Test for Two Binomial Proportions with a Log Relative Risk Statistic
The MODEL=TWOSAMPLEFREQ(TEST=LOGRR) option in the SAMPLESIZE statement derives the sample size required to test two binomial proportions by using a log relative risk statistic. The relative risk is the ratio of the proportion in one group to the proportion in the other group. The log relative risk statistic is the logarithm of the relative risk
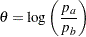 |
The hypothesis of no difference between two proportions, 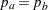 , can be tested through the null hypothesis
, can be tested through the null hypothesis 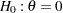 . For example, with
. For example, with 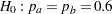 and
and  , it corresponds to the equivalent hypothesis
, it corresponds to the equivalent hypothesis 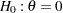 and
and 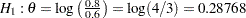 .
.
The maximum likelihood estimate of 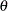 is given by
is given by
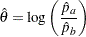 |
with an asymptotic variance
 |
where 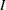 is the information (Chow and Liu 1998, p. 329).
is the information (Chow and Liu 1998, p. 329).
In practice, 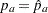 and
and 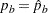 , the estimated sample proportions for groups A and B, respectively, at stage
, the estimated sample proportions for groups A and B, respectively, at stage 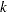 , are used to derive the information
, are used to derive the information 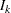 and the test statistic
and the test statistic 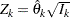 .
.
The maximum information 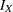 and proportions
and proportions  and
and 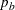 are needed to derive the required sample size. If the maximum information is not specified or derived with the ALTREF= option in the procedure, the PROP= option in the SAMPLESIZE statement is used to provide proportions under the alternative hypothesis for the alternative reference and then to derive the maximum information.
are needed to derive the required sample size. If the maximum information is not specified or derived with the ALTREF= option in the procedure, the PROP= option in the SAMPLESIZE statement is used to provide proportions under the alternative hypothesis for the alternative reference and then to derive the maximum information.
Note that in order to derive the sample sizes 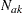 and
and 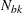 uniquely from the information,
uniquely from the information, 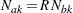 is assumed for
is assumed for 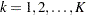 , where
, where  is the constant allocation ratio computed from the WEIGHT=
is the constant allocation ratio computed from the WEIGHT=
 option in the SAMPLESIZE statement. Then the sample size can be computed from
option in the SAMPLESIZE statement. Then the sample size can be computed from
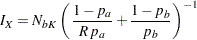 |
In PROC SEQDESIGN, the computed sample sizes in the two groups are
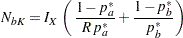 |
 |
where  is the constant allocation ratio, and
is the constant allocation ratio, and 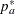 and
and 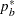 are proportions specified with the REF= option:
are proportions specified with the REF= option:
REF=NULLPROP uses proportions under
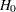 :
:  ,
, 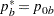
REF=AVGNULLPROP uses the average proportion under
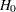 :
: 
REF=PROP uses proportions under
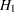 :
: 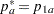 ,
, 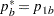
REF=AVGPROP uses the average proportion under
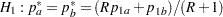
You can specify the MODEL=TWOSAMPLEFREQ( TEST=LOGRR WEIGHT=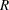 ) option in the SAMPLESIZE statement to compute the required total sample size and individual sample size at each stage. A procedure such as PROC LOGISTIC can be used to derive the log relative risk statistic.
) option in the SAMPLESIZE statement to compute the required total sample size and individual sample size at each stage. A procedure such as PROC LOGISTIC can be used to derive the log relative risk statistic.
Test for Two Survival Distributions with a Log-Rank Test
The MODEL=TWOSAMPLESURV option in the SAMPLESIZE statement derives the number of events required for a log-rank test of two survival distributions. The analysis of survival data involves the survival times for both censored and uncensored data. A noncensored survival time is the time from treatment to an event such as remission or relapse for an individual. A censored survival time is the time from treatment to the time of analysis for an individual surviving at that time, and the status is unknown beyond that time.
Let 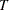 be the random variable of the survival time. Then the survival function
be the random variable of the survival time. Then the survival function
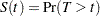 |
is the probability that an individual from the population has a survival time that exceeds  . And the hazard function is given by
. And the hazard function is given by
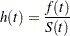 |
where 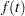 is the density function of
is the density function of 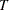 .
.
The hazard functions can be used to test the equality of two survival distributions  with the null hypothesis
with the null hypothesis 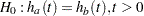 , where
, where 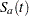 and
and 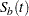 are survival functions for groups A and B, respectively, and
are survival functions for groups A and B, respectively, and 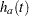 and
and 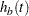 are the corresponding hazard functions.
are the corresponding hazard functions.
If the two hazards are proportional, 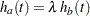 , where
, where 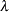 is a constant, then an equivalent null hypothesis is
is a constant, then an equivalent null hypothesis is
 |
Alternatively, another equivalent null hypothesis is given by
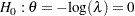 |
Suppose that the hazard rate  is a constant. Then with a specified median survival time
is a constant. Then with a specified median survival time 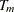 , the hazard rate can be derived from the equation
, the hazard rate can be derived from the equation
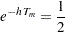 |
Denote the distinct event times at stage 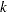 as
as  , where
, where  is the total number of distinct event times. Then the score statistic is the log-rank statistic (Jennison and Turnbull 2000, pp. 259–261; Whitehead 1997, pp. 36–39)
is the total number of distinct event times. Then the score statistic is the log-rank statistic (Jennison and Turnbull 2000, pp. 259–261; Whitehead 1997, pp. 36–39)
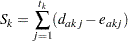 |
where 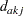 is the number of events from group A and
is the number of events from group A and 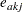 is the number of expected events from A. The number of expected events from
is the number of expected events from A. The number of expected events from  is computed as
is computed as
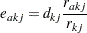 |
where 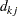 is the number of events from both groups,
is the number of events from both groups,  is the number of individuals from the treatment group who survived up to time
is the number of individuals from the treatment group who survived up to time 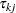 , and
, and 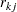 is the number of individuals from both groups who survived up to time
is the number of individuals from both groups who survived up to time 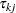 .
.
If the number of events 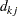 is small relative to
is small relative to 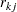 , the number of individuals survived up to time
, the number of individuals survived up to time 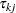 , then with a sufficiently large sample size,
, then with a sufficiently large sample size, 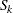 has an approximately normal distribution
has an approximately normal distribution
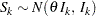 |
where the variance of 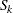 is the estimated information
is the estimated information
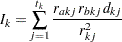 |
In order to derive the number of events from the information 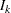 ,
, 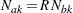 is assumed for
is assumed for 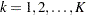 , where
, where  is the constant allocation ratio computed from the WEIGHT=
is the constant allocation ratio computed from the WEIGHT=
 option in the SAMPLESIZE statement.
option in the SAMPLESIZE statement.
The maximum information 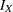 is needed to derive the required sample size. If the maximum information is specified or derived with the ALTREF= option in the procedure, the HAZARD=, MEDSURVTIME=, and HAZARDRATIO= options are not applicable. Otherwise, the HAZARD=, MEDSURVTIME=, or HAZARDRATIO= option is used to compute the alternative reference and then to derive the maximum information for the sample size calculation.
is needed to derive the required sample size. If the maximum information is specified or derived with the ALTREF= option in the procedure, the HAZARD=, MEDSURVTIME=, and HAZARDRATIO= options are not applicable. Otherwise, the HAZARD=, MEDSURVTIME=, or HAZARDRATIO= option is used to compute the alternative reference and then to derive the maximum information for the sample size calculation.
With 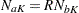 , if the number of events is few relative to the number of individuals who survived, then
, if the number of events is few relative to the number of individuals who survived, then  , and
, and
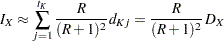 |
where 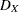 is the total number of events.
is the total number of events.
Thus, the required total number of events
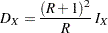 |
For a study group, if the hazard rate is constant, corresponding to an exponential survival distribution, and the individual accrual is uniform in the accrual time  with a constant accrual rate
with a constant accrual rate 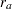 , then the required total sample size and sample size at each stage can be derived. See the section Input Number of Events for Fixed-Sample Design for a detailed description of the sample size computation that uses hazard rates, accrual rate, and accrual time.
, then the required total sample size and sample size at each stage can be derived. See the section Input Number of Events for Fixed-Sample Design for a detailed description of the sample size computation that uses hazard rates, accrual rate, and accrual time.
You can specify the MODEL=TWOSAMPLESURVIVAL option in the SAMPLESIZE statement to compute the required total number of events and individual number of events at each stage. With the specifications of hazard rates, accrual rate, and accrual time, the required total sample size and individual sample size at each stage can also be derived. If the REF=NULLHAZARD option is specified, the hazard rates under the null hypothesis, 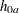 and
and 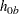 , are used in the sample size computation. Otherwise, the hazard rates under the alternative hypothesis,
, are used in the sample size computation. Otherwise, the hazard rates under the alternative hypothesis, 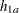 and
and  , are used. A procedure such as PROC LIFETEST can be used to derive the log-rank statistic.
, are used. A procedure such as PROC LIFETEST can be used to derive the log-rank statistic.
Copyright © SAS Institute, Inc. All Rights Reserved.