| The FREQ Procedure |
Example 3.7 Cochran-Mantel-Haenszel Statistics
The data set Migraine contains hypothetical data for a clinical trial of migraine treatment. Subjects of both genders receive either a new drug therapy or a placebo. Their response to treatment is coded as 'Better' or 'Same'. The data are recorded as cell counts, and the number of subjects for each treatment and response combination is recorded in the variable Count.
data Migraine; input Gender $ Treatment $ Response $ Count @@; datalines; female Active Better 16 female Active Same 11 female Placebo Better 5 female Placebo Same 20 male Active Better 12 male Active Same 16 male Placebo Better 7 male Placebo Same 19 ;
The following PROC FREQ statements create a multiway table stratified by Gender, where Treatment forms the rows and Response forms the columns. The CMH option produces the Cochran-Mantel-Haenszel statistics. For this stratified 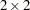 table, estimates of the common relative risk and the Breslow-Day test for homogeneity of the odds ratios are also displayed. The NOPRINT option suppresses the display of the contingency tables. These statements produce Output 3.7.1 through Output 3.7.3.
table, estimates of the common relative risk and the Breslow-Day test for homogeneity of the odds ratios are also displayed. The NOPRINT option suppresses the display of the contingency tables. These statements produce Output 3.7.1 through Output 3.7.3.
proc freq data=Migraine; tables Gender*Treatment*Response / cmh; weight Count; title 'Clinical Trial for Treatment of Migraine Headaches'; run;
| Cochran-Mantel-Haenszel Statistics (Based on Table Scores) | ||||
|---|---|---|---|---|
| Statistic | Alternative Hypothesis | DF | Value | Prob |
| 1 | Nonzero Correlation | 1 | 8.3052 | 0.0040 |
| 2 | Row Mean Scores Differ | 1 | 8.3052 | 0.0040 |
| 3 | General Association | 1 | 8.3052 | 0.0040 |
For a stratified 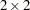 table, the three CMH statistics displayed in Output 3.7.1 test the same hypothesis. The significant
table, the three CMH statistics displayed in Output 3.7.1 test the same hypothesis. The significant 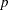 -value (0.004) indicates that the association between treatment and response remains strong after adjusting for gender.
-value (0.004) indicates that the association between treatment and response remains strong after adjusting for gender.
The CMH option also produces a table of relative risks, as shown in Output 3.7.2. Because this is a prospective study, the relative risk estimate assesses the effectiveness of the new drug; the "Cohort (Col1 Risk)" values are the appropriate estimates for the first column (the risk of improvement). The probability of migraine improvement with the new drug is just over two times the probability of improvement with the placebo.
The large 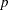 -value for the Breslow-Day test (0.2218) in Output 3.7.3 indicates no significant gender difference in the odds ratios.
-value for the Breslow-Day test (0.2218) in Output 3.7.3 indicates no significant gender difference in the odds ratios.
| Estimates of the Common Relative Risk (Row1/Row2) | ||||
|---|---|---|---|---|
| Type of Study | Method | Value | 95% Confidence Limits | |
| Case-Control | Mantel-Haenszel | 3.3132 | 1.4456 | 7.5934 |
| (Odds Ratio) | Logit | 3.2941 | 1.4182 | 7.6515 |
| Cohort | Mantel-Haenszel | 2.1636 | 1.2336 | 3.7948 |
| (Col1 Risk) | Logit | 2.1059 | 1.1951 | 3.7108 |
| Cohort | Mantel-Haenszel | 0.6420 | 0.4705 | 0.8761 |
| (Col2 Risk) | Logit | 0.6613 | 0.4852 | 0.9013 |
Copyright © SAS Institute, Inc. All Rights Reserved.